Note: This is a project under development. The articles on this wiki are just being initiated and broadly incomplete. You can Help creating new pages.
Temperature
A temperature [1] is an objective comparative measurement of hot or cold. It is measured by a thermometer. Several scales and units exist for measuring temperature, the most common being Celsius (denoted °C; formerly called centigrade), Fahrenheit (denoted °F), and, especially in science, Kelvin (denoted K).
The coldest theoretical temperature is absolute zero, at which the thermal motion of atoms and molecules reaches its minimum – classically, this would be a state of motionlessness, but quantum uncertainty dictates that the particles still possess a finite zero-point energy. Absolute zero is denoted as 0 K on the Kelvin scale, −273.15 °C on the Celsius scale, and −459.67 °F on the Fahrenheit scale.
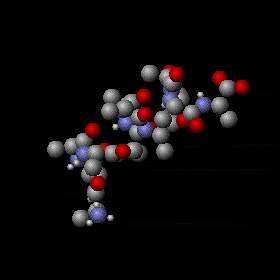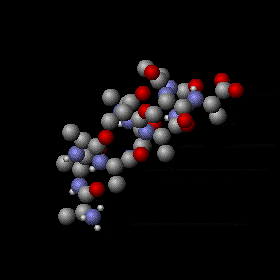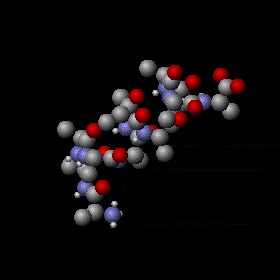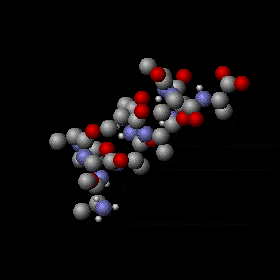
The kinetic theory offers a valuable but limited account of the behavior of the materials of macroscopic bodies, especially of fluids. It indicates the absolute temperature as proportional to the average kinetic energy of the random microscopic motions of those of their constituent microscopic particles, such as electrons, atoms, and molecules, that move freely within the material. Thermal vibration of a segment of protein alpha helix: The amplitude of the vibrations increases with temperature.
Temperature is important in all fields of natural science including physics, geology, chemistry, atmospheric sciences, medicine and biology as well as most aspects of daily life.